Chemical Reactions And Equations Notes for Class 10 Science Chapter 1 – 2025-26
Class 10 Science Chapter 1 – Chemical Reactions and Equations Notes for CBSE session 2025–26 by Vidhyayan Academy are designed to make learning simple, effective, and exam-ready. These chemical reactions and equations notes break down complex concepts into easy, point-wise explanations so that students can grasp fundamentals quickly. With these chemical reactions and equations notes class 10, learners will not only understand how to balance equations step by step but also revise key concepts like the law of conservation of mass and the mole concept. Covering all important topics from Class 10 Science Chemical Reactions and Equations, these notes serve as a perfect resource for revision. If you are looking for comprehensive Class 10 Science Chemical Reactions and Equations Notes, this study material will help you prepare smartly and score better in exams.
At Vidyayan Academy, the focus is on providing easy-to-read Chemical Reactions And Equations Notes that highlight all the essential points and quick summaries, making revision stress-free and efficient. These Chemical Reactions And Equations Science Notes not only strengthen conceptual understanding but also help students save time while preparing for exams. With structured content, simplified language, and exam-focused coverage, these notes serve as the perfect revision companion for scoring high marks.
Table of Contents
1.1 Introduction – Chemical Reactions And Equations Notes
If we look closely at the world around us, it’s easy to notice that change is happening all the time. Some of these changes are simple and temporary, like ice melting into water or water turning into steam. In such cases, the substance remains the same, only its form or state changes. But there are many other changes where something entirely new is created, and the original material can never be brought back in its earlier form. These are called chemical changes, and the process behind them is known as a chemical reaction. To understand such processes in a structured way, students often refer to Chemical Reactions and Equations Notes, which explain how substances interact, transform, and form new products.

Think about everyday situations. On a hot summer day, if milk is left outside, it soon turns sour as bacteria act on it and form lactic acid. The milk has now become curd — a completely new substance. Similarly, when an iron nail or pan is left in moist air, it slowly develops a reddish-brown layer called rust. Grapes kept for a long time ferment and change into alcohol, and when we cook food, the raw ingredients are transformed into something tastier and easier to digest. Even inside our body, thousands of chemical reactions take place every second. The digestion of food and the process of respiration, where glucose breaks down in the presence of oxygen to release energy, are both perfect examples. All these changes are explained in detail in Chemical Reactions and Equations Notes for Class 10, making it easier for students to understand the importance of Class 10 Science Chemical Reactions and Equations in daily life
In all these cases, the original substance loses its identity and new products are formed. This is what makes Chemical Reactions And Equations Notes so important — they are not limited to laboratories or industries, but are happening constantly in nature and in our own lives.
Watch Our Animated Explanation Video
To make learning easier and more engaging, we’ve created an animated explanation video on Class 10 Chapter – Chemical Reactions and Equations notes. This video is available on the Vidhyayan Academy YouTube Channel, where concepts are explained step by step with visuals, examples, and simple language. Don’t just read the notes—watch the animation and understand how chemical reactions happen in real life. It will help you remember better and score higher in exams!
Watch Our Animated Explanation Video: Click Here
Also visit:
- Mind Map Class 6 Science Chapter 1 – The Wonderful World of Science
- How to Create the Perfect Study Timetable for Class 10 Students | The Pomodoro Technique
- How Do Airplanes Fly? Must know Physics Behind it.
- 5 Proven Group Discussion Skills for Student Success.
1.2 Characteristics of Chemical Reactions
In this Chemical Reactions and Equations notes for class 10 we will understand that Whenever a chemical reaction takes place, it often leaves behind some clear signs that help us recognize that a change has actually occurred. These signs are not just random; they are reliable indicators that new substances with different properties are being formed.
- Evolution of Gas
In some chemical reactions, a gas is released as a product. For example,
- When you add zinc granules to dilute sulphuric acid, you will immediately notice bubbles forming. These bubbles are hydrogen gas being released during the reaction. Similarly, when baking soda reacts with vinegar, carbon dioxide gas is produced.
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g) ↑
- Change in Colour
Certain reactions are identified by a visible change in colour.
- When citric acid reacts with purple-coloured potassium permanganate solution, the colour changes from purple to colourless.
- When an iron nail is dipped in a blue solution of copper sulphate. After some time, the blue colour fades and a greenish solution of iron sulphate appears, while the nail gets coated with reddish-brown copper. The change in colour shows that a new substance has been formed.
- Similarly, the reaction of sulphur dioxide gas with acidified potassium dichromate solution results in a colour change from orange to green.
- Change in State of Substance
Some reactions involve a transformation in the state of matter. .
- the combustion of candle wax shows a change in state—wax (solid) melts into liquid, while burning produces water (liquid) and carbon dioxide (gas). Such reactions may also display more than one characteristic at the same time.
4. Change in Temperature
Many reactions are accompanied by a rise or fall in temperature
- When quicklime reacts with water to form slaked lime, the reaction releases heat, causing a rise in temperature.
- Similarly, zinc granules reacting with dilute sulphuric acid also cause a noticeable rise in temperature
- Formation of Precipitate
In some reactions, an insoluble solid (precipitate) is formed. For example, when barium chloride solution reacts with sulphuric acid, a white precipitate of barium sulphate is produced.
BaCl2(aq) + H2SO4(aq) → BaSO4(s) (ppt) + 2HCl(aq)
Thus, chemical reactions can usually be identified by these observable changes: a shift in state, a visible change in colour, the release of a gas, or a noticeable change in temperature. Together, these signs help us distinguish chemical reactions from ordinary physical changes.
1.3 Chemical Equations
(a) Word Equation
When we describe a Chemical Reactions and Equations notes in words, the explanation often becomes long and repetitive. To make things simpler, scientists use a special way of writing reactions known as a chemical equation. A chemical equation is a short and precise representation of what happens in a reaction, showing the reactants (the starting substances) and the products (the new substances formed). In this Chemical Reactions and Equations Notes we know it better.
(b) Skeletal Equation
Instead of words, we can use symbols and formulas of the substances. Magnesium is written as Mg, oxygen as O₂, and magnesium oxide as MgO. So, the equation becomes:
Mg + O₂ → MgO
Take a simple example. When magnesium burns in oxygen, it produces a white powder called magnesium oxide. In words, this is written as:
Magnesium + Oxygen → Magnesium oxide
(c) Balanced Chemical Equation
A balanced chemical equation has the number of atoms of each element equal on both sides. Since the law of conservation of mass tells us that matter can neither be created nor destroyed, we must make sure that the number of atoms of each element is equal on both sides. To do this, we balance the equation. For example:
Zn + H₂SO₄ → ZnSO₄ + H₂
Here, the number of atoms is already equal, so it is a balanced equation.
1.4 Steps to Balance a Chemical Equation
Balancing a chemical equation means making sure the number of atoms of each element is the same on both sides of the reaction. Here in Chemical Reactions and Equations Notes along with balanced equation.
Step 1: Write the unbalanced equation
- Start by writing the correct formulas of reactants and products.
- Example: Fe+H2O→Fe3O4+H2Fe + H_2O \rightarrow Fe_3O_4 + H_2Fe+H2O→Fe3O4+H2
Step 2: Count the atoms on both sides
- List the number of atoms for each element in reactants and products.
- Example:
- Left: Fe = 1, H = 2, O = 1
- Right: Fe = 3, H = 2, O = 4
Step 3: Balance one element at a time
- Start with the element that appears in the most complex compound.
- Balance oxygen first by putting a coefficient 4 before H₂O.
Fe+4H2O→Fe3O4+H2Fe + 4H_2O \rightarrow Fe_3O_4 + H_2Fe+4H2O→Fe3O4+H2
Step 4: Balance hydrogen atoms
- Left: 4×2 = 8 H
- Right: 2 → so put 4 before H₂.
Fe+4H2O→Fe3O4+4H2Fe + 4H_2O \rightarrow Fe_3O_4 + 4H_2Fe+4H2O→Fe3O4+4H2
Step 5: Balance metal atoms (Fe here)
- Left: Fe = 1
- Right: Fe = 3 → put 3 before Fe.
3Fe+4H2O→Fe3O4+4H23Fe + 4H_2O \rightarrow Fe_3O_4 + 4H_23Fe+4H2O→Fe3O4+4H2
Step 6: Double-check all atoms
- Fe: 3 = 3
- O: 4 = 4
- H: 8 = 8
Balanced!
To Make Equations More Informative:
Writing the symbols of physical states of substances in a chemical equation:
By writing the physical states of substances, a chemical equation becomes more informative.
• For solids, the symbol is “(s)”.
• For liquids, it is “(l)”.
• For gases, it is “(g)”.
• For aqueous solutions, it is “(aq)”.
• For gas produced in the reaction, it is represented by “(↑)”.
• For precipitate formed in the reaction, it is represented by “(↓)”.
A balanced chemical equation gives complete information about a reaction — it tells us the reactants, the products, their proportions, and sometimes even the physical states (solid, liquid, gas, or aqueous). This makes chemical equations not only shorter but also more meaningful than simple word descriptions.
1.5 Types of Chemical Reactions
Chemical reactions are broadly classified into five main types. Let’s go one by one ini Chemical Reactions and Equations Notes along with diagrams:
1. Combination Reaction
Definition:
A reaction in which two or more reactants combine to form a single product is called a combination reaction.
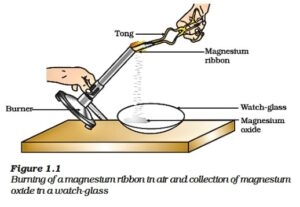
General form: A general combination reaction can be represented by the chemical equation given here: A + B → AB
Examples:
When magnesium is burnt in the air (oxygen), magnesium oxide is formed. In this reaction, magnesium is combined with oxygen.
Mg(s) + O2(g) → 2MgO(s)
Magnesium + Oxygen → Magnesium Oxide
2. Decomposition Reaction
Definition:
A reaction in which a single compound breaks down into two or more simpler substances (elements or compounds), usually by heat, light, or electricity, is called a decomposition reaction.
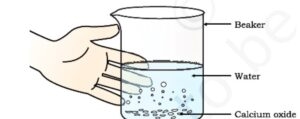
A general decomposition reaction can be represented as follows :
AB → A + B
Examples:
When calcium carbonate is heated, it decomposes into calcium oxide and carbon dioxide.
CaCO3(s) heat−→− CaO(s) + CO2(g)
Calcium carbonate → Calcium oxide + Carbon dioxide
When ferric hydroxide is heated, it decomposes into ferric oxide and water
2Fe(OH)3(s) △→ Fe2O3(s) + 3H2O(l)
3. Displacement Reaction
Definition:
A reaction in which a more reactive element displaces a less reactive element from its compound is called a displacement reaction.
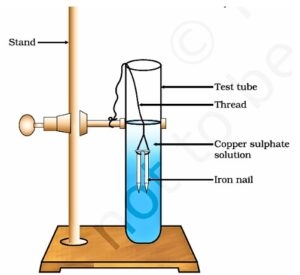
General form: AB + CD → AC + BD
Examples:
Fe(s)+CuSO4(aq)→FeSO4(aq)+Cu(s)
Here, iron (Fe) is more reactive than copper (Cu), so it displaces copper from copper sulphate solution, forming iron sulphate and depositing copper metal.
4. Double Displacement Reaction
Definition:
A reaction in which there is an exchange of ions between two compounds, forming two new compounds, is called a double displacement reaction.
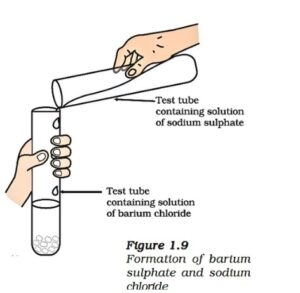
General form: AB+CD→AD+CB
Example: Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
Here, barium sulfate (BaSO₄) is formed as a precipitate.
5. Exothermic and Endothermic Reactions
These Chemical Reactions and Equations Notes for Class 10 explain that chemical reactions are often linked with changes in energy. In some reactions, energy is released, while in others, energy is absorbed. On this basis, reactions are classified into two types – exothermic and endothermic reactions.
1. Exothermic Reactions
- In these reactions, energy is released in the form of heat, light, or sound along with the products.
- As a result, the surroundings usually become warmer.
- Examples:
- Burning of fuel (combustion):
CH4+2O2→CO2+2H2O+HeatCH₄ + 2O₂ → CO₂ + 2H₂O + HeatCH4+2O2→CO2+2H2O+Heat
Natural gas, coal, or wood releases a large amount of heat when burnt. - Respiration:
Glucose in our body breaks down with oxygen to release carbon dioxide, water, and energy.
C6H12O6+6O2→6CO2+6H2O+EnergyC₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + EnergyC6H12O6+6O2→6CO2+6H2O+Energy
This energy keeps us alive and active. - Reaction of quicklime with water:
CaO+H2O→Ca(OH)2+HeatCaO + H₂O → Ca(OH)₂ + HeatCaO+H2O→Ca(OH)2+Heat
- Burning of fuel (combustion):
Key point: Exothermic reactions are very useful, as they provide energy for cooking, heating, transportation, and even biological processes like respiration.
2. Endothermic Reactions
- These are reactions in which energy is absorbed from the surroundings, usually in the form of heat, light, or electricity.
- As a result, the surroundings become cooler.
- Examples:
- Decomposition of calcium carbonate:
CaCO3→HeatCaO+CO2CaCO₃ \xrightarrow{Heat} CaO + CO₂CaCO3HeatCaO+CO2
Heat is absorbed to break down limestone. - Photosynthesis:
Plants absorb sunlight to convert carbon dioxide and water into glucose.
6CO2+6H2O→Sunlight,ChlorophyllC6H12O6+6O26CO₂ + 6H₂O \xrightarrow{Sunlight, Chlorophyll} C₆H₁₂O₆ + 6O₂6CO2+6H2OSunlight,ChlorophyllC6H12O6+6O2 - Electrolysis of water:
2H2O→Electricity2H2+O22H₂O \xrightarrow{Electricity} 2H₂ + O₂2H2OElectricity2H2+O2
- Decomposition of calcium carbonate:
Key point: Endothermic reactions are essential for processes like photosynthesis, which supports life on Earth.
6. Oxidation and Reduction (Redox Reaction)
Definition:
- Oxidation: Addition of oxygen or removal of hydrogen.
- Reduction: Addition of hydrogen or removal of oxygen.
When oxidation and reduction take place simultaneously in the same reaction, it is called a redox reaction.
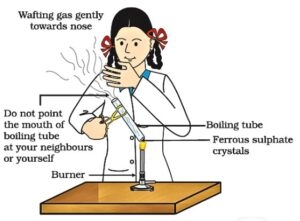
Example: CuO + H₂ → Cu + H₂O
- CuO → Cu (reduction, because oxygen is removed)
- H₂ → H₂O (oxidation, because oxygen is added)
1.6 Effects of Oxidation in Daily Life
Oxidation reactions are very common in our surroundings, and while some are useful, many create problems in daily life. Two important effects of oxidation are explained below:
- Corrosion of Metals
- When metals are exposed to air, moisture, or other chemicals, they slowly react and form undesirable compounds on their surface.
- Example: Rusting of Iron – Iron reacts with oxygen and moisture to form a reddish-brown flaky substance called rust. This weakens the metal over time and causes damage to bridges, railings, car bodies, and pipelines.
- Other metals also undergo corrosion: silver turns black due to the formation of silver sulphide, while copper develops a green layer of basic copper carbonate.
- Corrosion not only spoils the appearance of metals but also reduces their strength, leading to heavy financial losses. That is why we paint, oil, or galvanize iron articles to prevent rusting.
- Rancidity of Oils and Fats
- Fats and oils in food items easily get oxidized when exposed to air, especially in warm conditions.
- This oxidation changes their smell and taste, making the food unfit for consumption. For example, old chips or fried snacks often give a bad odour due to rancidity.
- To prevent this, food is often stored in airtight containers, antioxidants are added, or packets are flushed with inert gases like nitrogen to slow down oxidation.
In short:
- Corrosion damages metals and weakens structures.
- Rancidity spoils food by giving it a bad taste and smell.
Both are harmful effects of oxidation, and controlling them is essential for protecting both materials and food in our daily life.
Final Words
Mastering Class 10 Science Chemical Reactions and Equations becomes much easier with well-structured notes. The chemical reactions and equations notes provided here simplify every topic, from balancing equations to understanding different types of reactions. These chemical reactions and equations notes class 10 are designed to give you clarity, quick revision, and confidence for exams. If you are preparing for the CBSE 2025–26 session, rely on these Class 10 Science Chemical Reactions and Equations Notes by Vidhyayan Academy to strengthen your concepts and score higher.
Frequently Asked Questions (FAQs) on Chemical Reactions and Equations Notes
Q1. What is a chemical reaction?
A chemical reaction is a process in which one or more substances (reactants) are converted into new substances (products) with different properties.
Q2. What are the different types of chemical reactions?
The main types are:
- Combination reaction
- Decomposition reaction
- Displacement reaction
- Double displacement reaction
- Redox reaction
These are explained in detail in Chemical Reactions and Equations Notes for Class 10.
Q3. What is the difference between a physical change and a chemical change?
- In a physical change, only the state or form of a substance changes (like ice melting).
- In a chemical change, a completely new substance is formed (like rusting of iron).
Q4. Why is it important to balance chemical equations?
Balancing ensures that the law of conservation of mass is followed—atoms are neither created nor destroyed in a chemical reaction. This makes the equation scientifically correct.
Q5. How can I study Chemical Reactions and Equations easily?
The best way is to use Chemical Reactions and Equations Notes along with diagrams, balanced equations, and real-life examples. Watching animated videos also helps in quick understanding.
